

News and Events
аТЮХзЪбЖ
аТЮХзЪбЖ

2026-04-10
ШЋСДЬѕУ№ОњЃЌЛЄКНвћЦЗЧхОВЁЊЁЊЭўСЎЯЃЖћЙйЭјЕижЗЩњЮяаЏЙрзАЯпVHPМЦЛЎССЯрЩюлк CBST 2026
2026ФъЃЌCBSTжаЙњЙњМЪвћСЯЙЄвЕПЦММеЙдкЩюлкЪЂДѓЦєФЛ
ЁЃзїЮЊвћСЯЙЄвЕСьгђМЋОпгАЯьСІЕФзЈвЕЪЂЛсЃЌБОДЮеЙЛсЛуОлСЫШЋЧђвћСЯЙЄвЕСДЩЯЯТгЮЕФСЂвьЪжвеЁЂгХжЪзЪдДгыаавЕОЋгЂЃЌОлНЙвћСЯЩњВњШЋСїГЬЕФжЧФмЛЏЁЂЧхОВЛЏЩ§МЖЃЌЮЊЁ
ЩѓВщЯъЧщ

ЩѓВщЯъЧщ
272026.03
аТЦЗЩЯЪа | SS6-121RаЭПьЫйздКЌЪНбЙСІеєЦћУ№ОњЩњЮяжИЪОМСЃКИпаЇБуНнЃЌОЋзММрВт
дкЯжДњвНдКЯћЖОЙЉгІжааФжЪПиЙмРэЯЕЭГжаЃЌУ№ОњМрВтЪЧАќЙмвНСЦЧхОВЕФНЙЕуЛЗНк
ЁЃетвЛЛЗНкВЛЕЋвЊЧѓМрВтаЇЙћОЋзМПЩППЃЌИќашвЊИпаЇЕФСїГЬжЇГж
ЁЃЁАУ№ОњМАИёЁБвбВЛдйжЛЪЧвЛЯюЪ§ОнжИЁ

ЩѓВщЯъЧщ
252026.03
ИЩЛѕЫйЕн | УИЕзЮяЗЈДѓГІОњШКМьВтвЩФбЮЪЬтШЋРњГЬЦЪЮі
ДЫЧАЃЌЮвУЧвд3ЗжжгПЦЦеаЮЪНЦеМАСЫДѓГІОњШКЯрЙижЊЪЖЃЌЪеЕНжкЖргУЛЇЙигкМьВтЪЕВйЕФЬсЮЪЃЌР§ШчКЌТШЗЯЫЎЕФдЄДІжУГЭЗЃвЊЧѓЁЂИпгВЖШЫЎбљЖдаЇЙћЕФгАЯьЁЂЧГЩЋПзЕФХаЖСБъзМЕШ
ЁЃБОЦкФкШнОлЁ
ЩѓВщЯъЧщ
242026.03
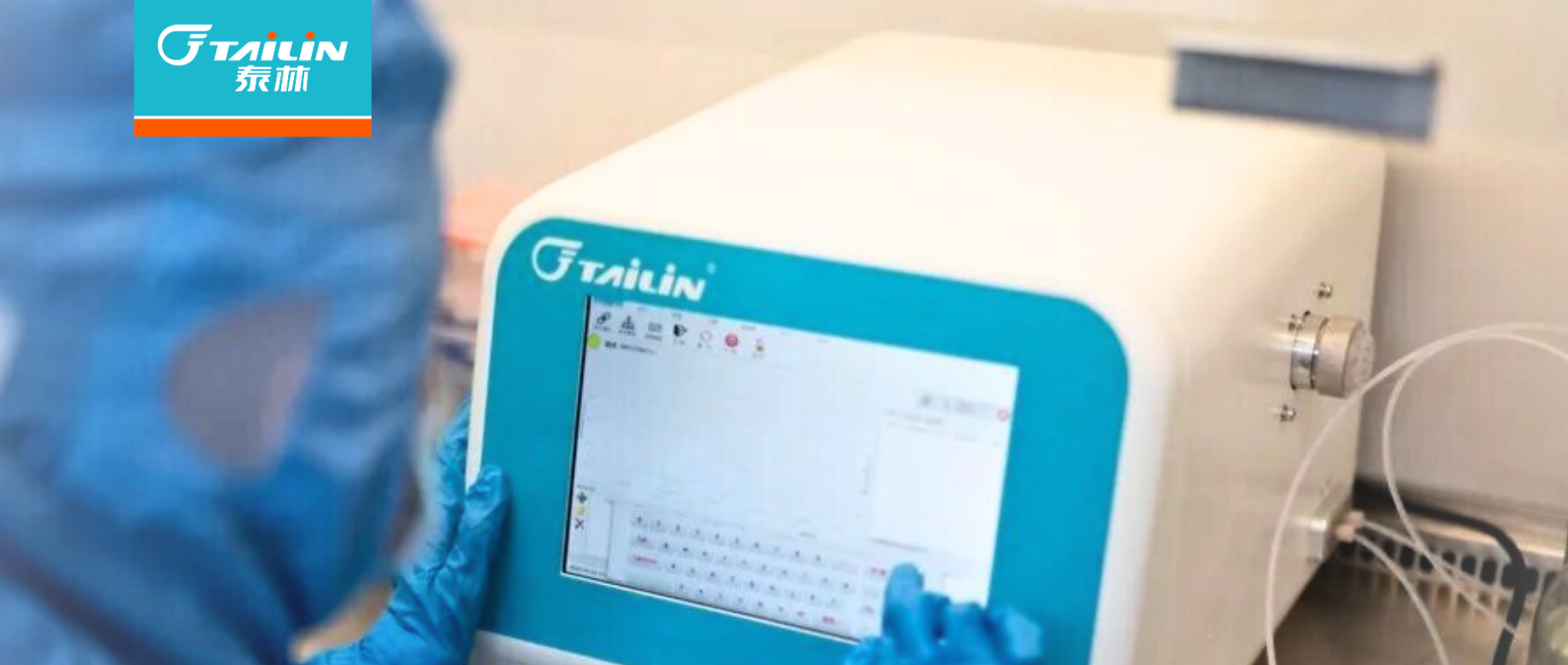
ШЈЭўШЯжЄ+жеЖЫШЯПЩЃЁЭўСЎЯЃЖћЙйЭјЕижЗMD500ЮЂЩњЮяПьМьвЧЃЌгУЪ§ОнжўРЮжЦвЉКЯЙцЗРЕи
ПЫШеЃЌеуНЭўСЎЯЃЖћЙйЭјЕижЗЦЪЮівЧЦїгаЯоЙЋЫОзджїбаЗЂЕФMD500аЭЮЂЩњЮяПьЫйМьВтЦЪЮівЧЃЌВЛЕЋЫГЫьЭЈЙ§еуНЪЁЪГЮявЉЦЗФЅСЗбаОПдКЃЈвдЯТМђГЦЁАЪЁвЉМьдКЁБЃЉЬцЛЛвЊСьбЇШЈЭўбщжЄЃЌИќНЋетЗнбщЁ
ЩѓВщЯъЧщ
132026.03
ЧГЮіЁЖGB/T 18281вНСЦБЃНЁВњЦЗУ№Оњ ЩњЮяжИЪОЮяЁЗЕФзЊБфгыЕїНт
ЁЖGB/T 18281вНСЦБЃНЁВњЦЗУ№Оњ ЩњЮяжИЪОЮяЁЗЪЧгаЙиЩњЮяжИЪОМСЕФЭЈгУадКЭзЈгУадБъзМ
ЁЃаТАцЁЖGB/T 18281-2024 вНСЦБЃНЁВњЦЗУ№Оњ ЩњЮяжИЪОЮяЁЗЯЕСаБъзМгк2024Фъ11дТ28ШеЁ

ЩѓВщЯъЧщ
042026.03
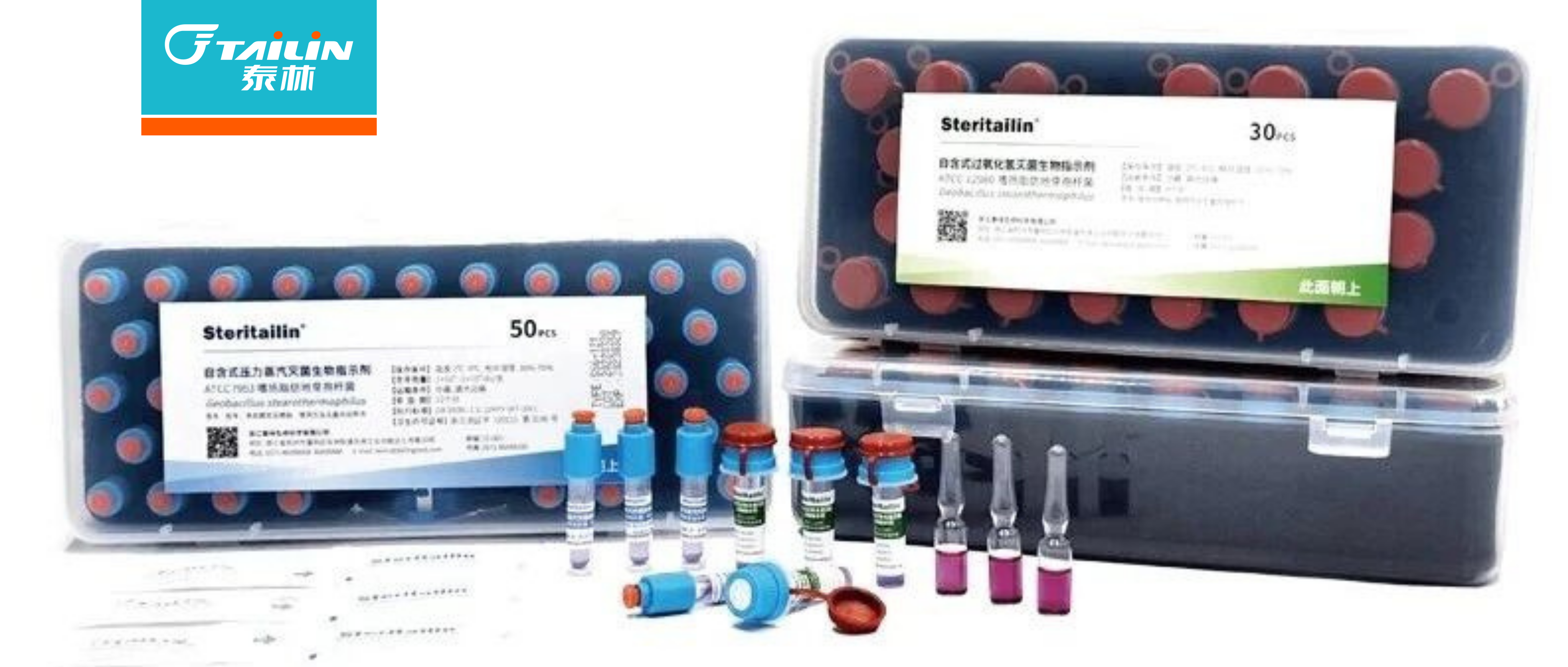
ЭўСЎЯЃЖћЙйЭјЕижЗ Immutailin? PVDFзЊгЁФЄаТЦЗЩЯЪа
дкТбАзжЪгыКЫЫсМьВтЦЪЮігІгУжаЃЌNC зЊгЁФЄЃЈЯѕЫсЯЫЮЌЫиФЄЃЉгыPVDF зЊгЁФЄЃЈОлЦЋЖўЗњввЯЉФЄЃЉЪЧСНЯюНЙЕуЕФЙЬЯржЇГжНщжЪ
ЁЃЫќУЧвРИНИїздЦцвьЕФадФмгХЪЦЃЌЦеБщгІгУгкЁ


 EN
EN
